If composting requires elevated oxygen levels, do we need to measure both oxygen and temperature to understand the process?
Composters frequently ask us if we use compost oxygen monitoring. It’s a reasonable question. Composting is an aerobic biological process, and microorganisms require oxygen to metabolize organic material and generate heat.
Commercial composting success is largely a function of understanding how to manage key performance indicators. With any complex biological process, there is more data than anyone could ever hope to measure, and more opportunities for improvement than anyone could hope to (or budget to) implement. The challenge is deciding which measurements actually improve operations.
And as a science-driven company, we love data. But we typically do not record and track oxygen concentrations in our compost systems. Here’s why:
Compost aeration rates are driven by heat removal more than oxygen supply. Tracking oxygen is not necessary.
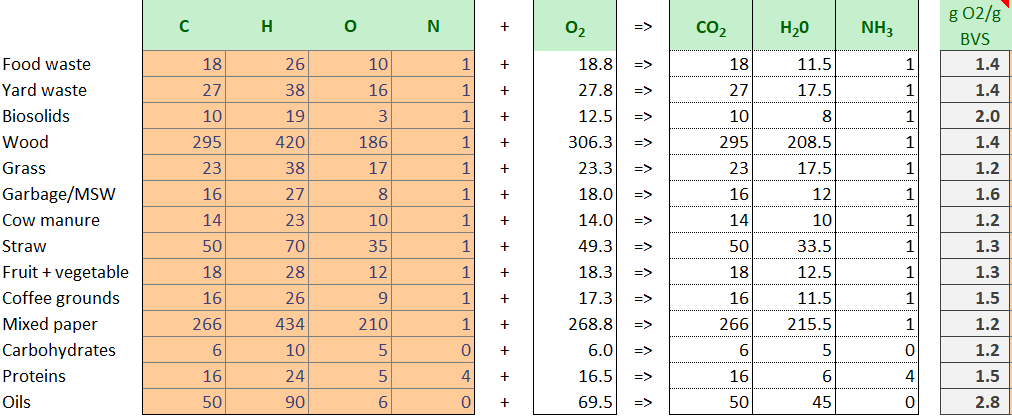
Composting is the biological process of aerobically decomposing organic compounds, composted of Carbon, Hydrogen, Oxygen and Nitrogen (C,H,O,N), to create primarily carbon dioxide and water. This C,H,O,N compound is commonly referred to as Bio-available Volatile Solids, or BVS.

We often refer to this microbial consumption and respiration process as bio-oxidation. And the faster the microbes bio-oxidize the goo, the faster we stabilize the compost. A couple minor notes regarding compounds that may form other than CO2, H2O, and NH3:
Bonding oxygen from the air to the organic matter through this process also releases considerable heat.
The following analysis explores and expands some examples provided in The Practical Handbook of Compost Engineering.
The table shows a variety of organic compounds found in composting. Stoichiometry involves looking at the quantity of reactants consumed and products produced in chemical reactions and their ratios. We use stoichiometry to calculate how much oxygen (O2) is required to balance the equation. Comparing the molar mass of the starting feedstock (BVS) vs the molar mass of required O2, we the ratio of oxygen per BVS. This is commonly in the 1.2-2.0 gram O2/ gram BVS range, though you can see oils require even more air. This is useful, because it tells us how much air is required to for composting based on stoichiometry.
For example, food waste may be represented as 18C + 26H + 10 O + 1N. Adding up the molar masses (18 * 12 + 26*1 + 10* 16 + 1*14)= 416. Molar wieght of 18.8 O2 = 18.8 * 16*2 = 600.
600/416 = 1.4 grams O2 per gram BVS.
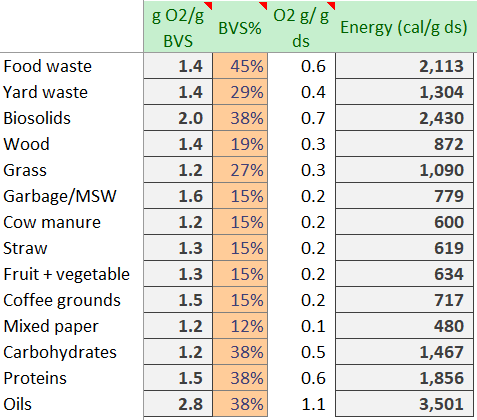
Calculating heat removal is a little more involved. At a fundamental level, when the oxygen bonds to carbon, it releases 20.5k calories per electron (2 electrons per oxygen, and 2 oxygens in O2), so 104 kcal per mol of O2. In a gram of O2, we have 104 * 1000/32 g/mol = 3,250 cal/g O2
Recognizing that much of inbound feedstock is water or ash, we can estimate the BVS fraction that will be oxidized. We multiply the oxygen ratio, by BVS%, and by 3,250 to determine the total heat released from the reaction.
For example, food waste might be as high as 45% BVS, so .45 * 3,250 = 2,113 calories per gram of dry solids. Since the O2 ratios minimally vary, the heat released is heavily impacted by the BVS% (which is an estimate based on the measured volatile solids and the estimated bio-available fraction of this).
To calculate the amount of air required, we have to make some assumptions on entering and exiting temperature, along with relative humidity. However, we quickly discover that even under a range of temperature conditions, the Excess Air Ratio (EAR) remains high (further down).
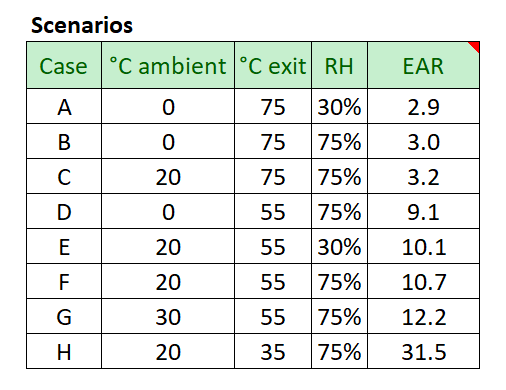
We can use psychrometrics to calculate the heat removal rate of air between two specific conditions. The first case assumes at 20°C (68°F) at 75%RH, with a 55°C (131°F) exhaust temperature, a common condition at many facilities.
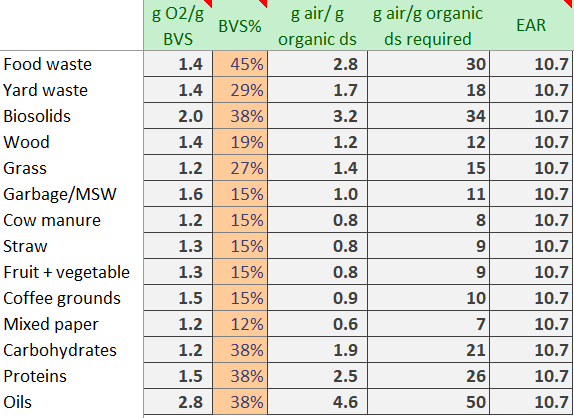
We see the mass of air is ~10-30x the weight of the mass of the organics to meet the cooling load (required grams of air per gram of organic dry solids ). Yikes. While front end loader, shredders, and trommels often catch our eye – it is the aeration fans quietly working in the background that are performing most of the material handling…air!
Comparing this air required for cooling vs the air required to complete the chemical reaction (ie 30 grams air vs 2.8 grams air for food waste), we calculate a consistent EAR of 10.7. This means that providing sufficient cooling requires over 10x the amount air required by the microbes to consume and compost the organics.
The EAR remains constant for different feedstocks, but will vary a small amount under different inlet and outlet air conditions. Check out some of the scenarios below. Notice how changing the exist air temperature (temerpature coming out of the pile) has a large impact on the EAR. This is because most of the energy removal comes from the latent heat of vaporization, converting liquid water to vapor. The exhaust air is normally saturated. At higher temperatures, air can transport much more water vapor, and thereby energy, which remove heat faster than the same mass flow of low temperature air.
Other variations have relatively small impact. Notice how modifying the relative humidity of the incoming air only affects the ratio by <1. Similarly modifying the ambient air temperature by 10°C on affects the EAR by ~10%.
The scenarios we modeled represent a wide range of potential climates and pile conditions. And even in extreme cases, we consistently see more air required for cooling than stoichiometry.
Temperature is a great key performance indicator. It’s relatively easy to use temperature probes, they have relatively small cost compared to other sensors, and they produce relatively representative data.
A caveat on the data – any massive organic pile will have variation from point to point. The temperature may be cooler and oxygen levels higher near where the air enters, for example. A further distance between temperature/oxygen measurement location and where the air enters will tend to have higher temperature and lower oxygen. The material density and porosity also plays a significant role. While this is often not a problem, improving mixing and shortening fill rates can help homogenize an aerated batch if needed. Improving mix porosity can improve airflow, which also helps.
With negative or reversing aeration, another approach that can work well is measuring the temperature of the exhaust air duct near the pile. While temperature insertion probes measure a small, local temperature, the exhaust air takes an average of the pile conditions. While there can be some thermal losses along the aeration floor and duct, this approach can provide a much more representative condition of the overall mass.
If the pile temperature rises, this indicates more heat generation than removal. If the pile temperature drops, we have more cooling than heat generation. A perfectly balanced system would maintain a steady state temperature.
However, a diverse pile of organic feedstocks with pockets of living microbes creating the heat – steady state is more of a conceptual idea than reality in the field. Modern compost control systems modulate airflow based on temperature feedback – seeking to maintain optimal temperature levels. We talk more about optimal temperatures as they relate to biodegradation and odor in this post. We find in the field and our lab that adequately controlled compost systems maintain about 18-20% O2 in the pore space (compared to ~23% O2 in outside air).
An adequately designed system should be able to cool an energetic pile in a reasonable timeframe (say 8-24 hours) under normal loading and design conditions. If the aeration is unable to provide reasonable cooling (several degrees) within this timeframe – the high temperatures are likely to inhibit the biological process.
Conversely, a system that can rapidly cool (which is very uncommon in commercial environments), may have more capacity than is currently utilized.
Composting systems can generate large amounts of data, but not every measurement leads to better decisions.
For most commercial systems, temperature monitoring provides the most actionable insight per sensor installed. With 10-11x more air required for heat removal than stoichiometry – heat removal firmly drives the process.
That doesn’t mean oxygen measurements have no value. They can be useful for research, troubleshooting, or validating system performance in specific situations.
But for day-to-day operations, controlling temperature and airflow typically ensures that oxygen levels remain well within the aerobic range.
And that’s why, in most cases, we focus our instrumentation on temperature first.
BVS – the bio-available volatile solids, also the degradable fraction of the volatile solids
g – grams, used for mass comparison in the calculations
cal – calories, used for the calculations
C: Carbon
H: hydrogen
O: Oxygen
N: nitrogen
RH: relative humidty